Let’s get back to our own science project.

The above chart shows two things we immediately recognize as very similar. In fact they seem to match each other very well. We would say they have a strong correlation, and with a little mathematics, we could compare each one point-by-point on the graph and come up with a number that would tell us just how well they match each other, called a correlation coefficient. In fact, a glance at the above suggests a perfect, 100% correlation, because in fact one is an exact copy of the other. We know this because we made the chart.
Now suppose the blue one represents changing CO2 levels in the atmosphere and the red one represents changing global temperatures over the same time frame. The above is a gross exaggeration, of course, but we wanted to make sure no one doubted there is a strong correlation between the two. Is there any doubt that CO2 is causing the global temperature to change? Any doubt at all? None? Zero doubt, right?
Wrong In fact, the blue line is exactly one pixel on your screen ahead of the red line. We know that because we made the chart. You couldn’t tell that one was exactly like the other and actually leading it by one pixel without dragging out a powerful magnifying device and doing some very careful measurements and some pretty lengthy mathematics.
This was the fundamental mistake that Mann, Al Gore, the Oscar boys, the Nobel Committee, the IPCC, and just about everyone else on the planet made at the beginning. They immediately assumed, noticing that CO2 levels and global temperatures had a pretty good correlation, that CO2 was the culprit, and was causing global temperatures to rise. In fact, it appears it was just the opposite: rising global temperatures caused increased CO2 level in the atmosphere.
So where did the increasing CO2 come from? You can’t make CO2 out of Oxygen and Nitrogen… surely you’re pulling my leg!
Let’s do a little simple Chemistry, and figure out the molecular mass of the different atmospheric constituents. For this we go to the Periodic Table of the Elements, and find the atomic mass of Nitrogen, Carbon, Hydrogen, and Oxygen. Let’s forget about Argon, which is about 0.9% of the atmosphere, because it’s supposed to be CO2 that’s the evil stuff. To the nearest round number, Carbon = 12, Nitrogen = 14, Oxygen = 16, and lowly Hydrogen = 1. That’s based on the number of protons and neutrons in the nucleus of the atom. The Periodic Table will give a slightly different number, because of that binding energy ( which is a mass equivalent ) we talked about earlier.
So, what’s the approximate molecular mass of the different gasses? That’s simple addition:
| Water (H2O)1 +1+16 = 18 amu | Nitrogen (N2)14 + 14 = 28 amu | Oxygen (O2)16 + 16 = 32 amu | Carbon Dioxide (CO2)12 + 16 + 16 = 44 amu |
Remember, we’re rounding off to the nearest whole number, and amu means Atomic Mass Units.
Do you see something significant? Think like a scientist. Yes, CO2 is by far the heaviest of the major constituents, and the law of gravity applies to it as well. It sinks to the ground.. in fact, into the ground, and into the oceans, as well, because CO2 is very water-soluble and that’s what puts the fizz in Ginger Ale.
This doesn’t happen overnight. In fact, the winds and convection currents and such keep the air stirred up constantly, so it may take 100-150 years for the CO2 you are exhaling right now to make it back into mother earth, where most of it is currently locked up.
 |
|
Now our puzzle is complete, and we can visualize the whole thing.
1. The sun heats the earth, repository of most of the CO2 on the planet.
2. Some stored CO2 comes out by a process known as outgassing ( from the soil ) and the champagne effect ( from the oceans ). The oceans are by far the largest source.
3. Sloppy “scientists” see the warming, and the CO2, but overlook the changes in the sun, don’t see the fine differences in timing… and proceed to blame the increasing temperature on CO2 and mankind as the culprit in a classic knee-jerk reaction.
Funny, any 1st Grader would have told us that if we had asked them “What makes the earth warm, Susie?” Nobody ever said science had to be “hard”. You can demonstrate this with a simple kitchen experiment. Pour a glass of ginger ale, sit it on the table, and see how long it takes to go “flat” at room temperature. Now pour an equal glass into a pan and put it on the stove on low heat, then time how long it takes to go flat. That’s your homework experiment – to demonstrate that extra heat really releases CO2 a lot faster :-)>
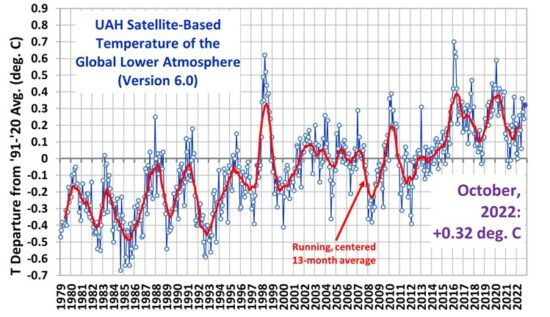
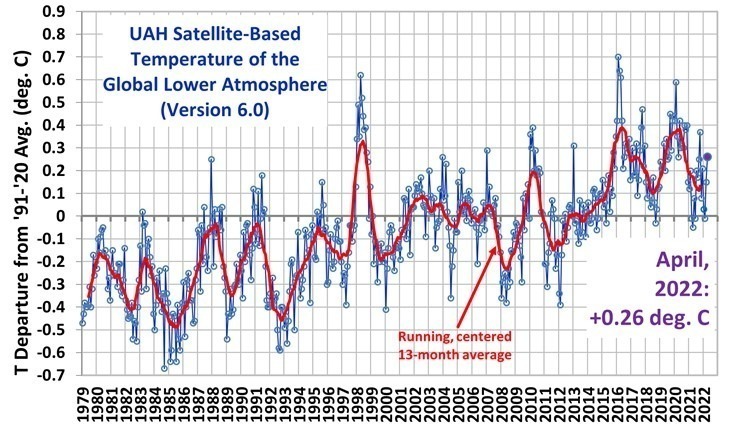
Our satellites are pretty good at measuring overall ocean temperatures from afar, and CO2 measurements are being taken daily around the globe. The best results we have been able to turn up so far is that measurable CO2 increases appear about 9 months after an upswing in ocean temperatures. The data is messed up a bit every time a volcano decides to blow its top, because that’s the mother of CO2 producers, bar none. And a buffalo emits about the same amount of methane (CH4) as driving your automobile about 8,000 miles – which can combine with O2 in a highly exothermic reaction ( gives off heat ) to produce CO2 and H2O as end products.
Continued at ‘Primer 7: Global Temperature’






On another miosisn I’ve just come across on what he might term denialist methodology. They don’t seem to care about the quality of the science they are putting up against AGW science as long as it creates doubt and confusion. Hence they use different papers in different contexts with equal enthusiasm, seemingly unaware that they contradict each other.Michael Tobis has found the right words: The science of AGW isn’t settled, but there is a coherent paradigm of well-established knowledge. New findings elaborate the paradigm, but we await something that will overturn it, which would be genuinely exciting. So far it hasn’t come.Gavin’s comment is also salutary:Many non-scientists have no clear idea of how highly rated redundancy is in science. You don’t really believe anything seriously before it has come from several independent sources. And those sources themselves are often internally redundant, like surface temperatures, monthly averaged, correlate over long distances.
Hi Fatima, thanks for your comment.
If you have studied my blog at all, you will realise that it is impossible for me to agree with what you say.
Here, and on relevant linked blogs, you will find genuine scientific evidence that, at the very least, establishes genuine doubt about the AGW claims.
I realize that the evidence is often at a technical level and complete understanding may sometimes be difficult, but people must think and judge for themselves.
Do not just accept other people’s conclusions, think about the evidence.
Maybe check out the WUWT website, where the evidence is vast and overwhelming.