Free Energy is high up on my list of interests and global energy resource.

The processing and eventual production of electrical energy is, of course, not free of costs, but it is certainly free of pollution and is 100% sustainable. Even though the author of this ‘Natural News‘ article suggests using the hydrogen for a more productive fusion process, there would be no necessity to go there, if we accept pollution free burning and steam turbine generators as adequate. Going ‘there’ involves incredible heat processes and, not sure about this but, ionising radiation?.
From Mike Adams, discussion on some hope for a cleaner future:
(NaturalNews) A team of scientists at the University of Colorado, Boulder, have achieved what appears to be the “holy grail” of water splitting technology for the production and storage of clean, abundant energy. Because sunlight is free, I’m calling this “free energy.”
To understand this breakthrough, it’s important to first understand why solar power has so many limitations. Solar is great when the sun is shining, but storing solar power require the deployment of a large array of heavy, expensive and toxic electrical storage devices known as “deep cycle batteries.” To put it in street terms, deep cycle battery technology sucks. The batteries suck, the chemicals suck, the weight sucks and the cost sucks. There is absolutely nothing to like about batteries unless you enjoy hulking around with heavy, useless objects.
So the “holy grail” of solar power has always been finding a way to store solar energy that’s portable, dense and relatively lightweight. Until now, that discovery has been elusive.
But now a team of scientists in Boulder, Colorado say they have come up with “a radically new technique that uses the power of sunlight to efficiently split water into its components of hydrogen and oxygen, paving the way for the broad use of hydrogen as a clean, green fuel.”
Sunlight, mirrors and a reaction chamber
The system works by exploiting a large array of ground mirrors to focus sunlight onto a tall reaction tower. There, the intense heat (2,500 degrees Fahrenheit) powers a reaction chamber containing metal oxides. The heat drives oxygen atoms off the metal oxides, causing them to “soak up” the oxygen from steam vapor introduced into the chamber. Steam vapor is, of course, made of water (H2O), so stealing the oxygen atoms from water leaves hydrogen gas that can then be collected.
In effect, the tower uses sunlight to split water into hydrogen gas and oxygen gas. The hydrogen gas is then collected, purified and pumped into high-pressure hydrogen containers which, pound for pound, are extremely dense “batteries” of energy that far out-perform chemical batteries.
Better yet, hydrogen gas then holds all this energy with 100% efficiency, losing no potential whatsoever, even if stored for decades. From an environmental perspective, hydrogen is also a super clean-burning fuel, producing no carbon dioxide emissions or particulate matter. (The reason hydrogen does not produce CO2 when combusted is because it does not contain carbon, obviously. CO2 production requires a carbon-based source of fuel such as hydrocarbons — coal, gasoline, diesel, etc.)
Read the complete article here.

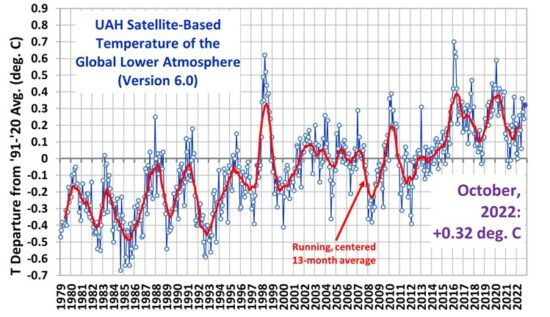



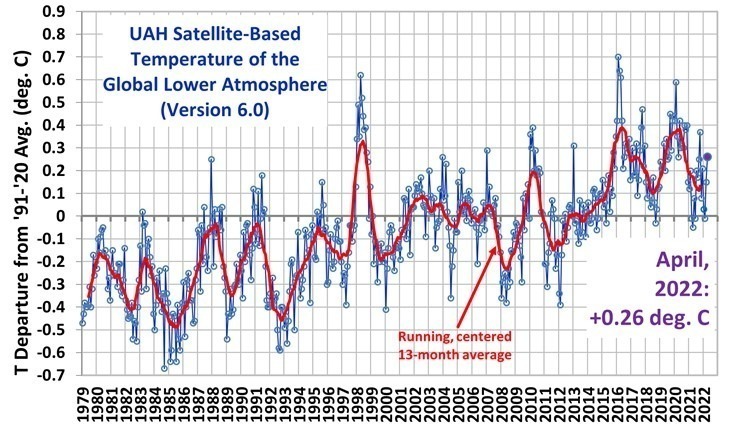


Thanks for the information.
I hope this contraption actually works.
Pingback: These items caught my eye – 4 August 2013 | grumpydenier
Thanks!