
English: Annual mean sea surface salinity from the World Ocean Atlas 2005. Salinity here is in practical salinity units (PSU). It is plotted here using a Mollweide projection (using MATLAB and the M_Map package). (Photo credit: Wikipedia)
I am moved to publish this commentary on the criticality of ocean parameters and effects on sea life, particularly oysters in this case.
THE MOTIVATION – Oysters Killed by Climate Change’s Evil Twin
The oyster industry is in trouble. A few years ago, oyster hatcheries in the Pacific Northwest began losing their “seed,” as oyster larvae are known, by the millions. They scrambled to figure out what was behind the massive die-off and discovered that it had to do with “ocean acidification,” also known as climate change’s evil twin.
As with climate change, ocean acidification happens when there’s too much carbon dioxide in the atmosphere. The earth’s waters absorb some of that CO2 — more than a quarter of the amount that we produce — as a way of achieving chemical equilibrium between water and air. “When CO2 mixes with seawater,” Jim Meyer explains, “it creates carbonic acid, and, spoiler alert, increasing acid in the oceans makes the oceans more acidic.”
As in nearly all issues of man-made CO2 influences on our lives, this particular area is not without counter-claims of bias and exaggeration.
I offer the following notes and publish them without editing or extra, deserved research just to get the ball rolling. I suggest the ‘jury is out’ the claim is devoid of credibility, to say the least! (Minor editing now employed).
Note that CO2 is required for ocean plants which in turn photosynthesize O2 which is necessary for the underwater animals/fish for respiration.
Also note that a liquid that is alkaline only becomes acidic when the pH is < 7.0, and the ocean is > 8.0. Technically, the ocean becomes more neutral, not more acidic! That is the start of the brainwashing.
Given the update information, it would have to be concluded that if a reduction in salinity is a problem it surely will not be due to the minor possible CO2 effect. It can only be a real acidic influence from a real pollutant.
GENERAL INFO:
Update information (Rev#1) – Very Relevant. Reference http://en.wikipedia.org/wiki/Oyster_farming
Cultivation

Oyster farming boats in Morbihan (Brittany, France)
Oysters naturally grow in estuarine bodies of brackish water. When farmed, the temperature and salinity of the water are controlled (or at least monitored), so as to induce spawning and fertilization, as well as to speed the rate of maturation – which can take several years.
From wiki on “brackish water” – “Brackish water or briny water is water that has more salinity than fresh water, but not as much as seawater.” And we are being asked to believe that minor variations in normal seawater are seriously detrimental!!!
Salinity
Salinity Map http://www.marinebio.net/marinescience/02ocean/swimg/salinity.jpg

Salinity map showing areas of high salinity (36 o/oo) in green, medium salinity in blue (35 o/oo), and low salinity (34 o/oo) in purple. Salinity is rather stable but areas in the North Atlantic, South Atlantic, South Pacific, Indian Ocean, Arabian Sea, Red Sea, and Mediterranean Sea tend to be a little high (green). Areas near Antarctica, the Arctic Ocean, Southeast Asia, and the West Coast of North and Central America tend to be a little low (purple).
The salinity of seawater is usually 35 parts per thousand (also written as o/oo) in most marine areas. This salinity measurement is a total of all the salts that are dissolved in the water. Although 35 parts per thousand is not very concentrated (the same as 3.5 parts per hundred, o/o, or percent) the water in the oceans tastes very salty. The interesting thing about this dissolved salt is that it is always made up of the same types of salts and they are always in the same proportion to each other (even if the salinity is different than average). The majority of the salt is the same as table salt (sodium chloride) but there are other salts as well. The table below shows these proportions: [The ‘carbon/”acid” part shown below].
Chemical Ion Contributing to Seawater Salinity: Bicarbonate
Concentration in o/oo (parts per thousand) in average seawater : 0.145
Proportion of Total Salinity (%) (no matter what the salinity) 0.41
Dissolved Gases
The concentration of dissolved oxygen and carbon dioxide are very important for marine life forms. Although both oxygen and carbon dioxide are a gas when outside the water, they dissolve to a certain extent in liquid seawater. Dissolved oxygen is what animals with gills use for respiration (their gills extract the dissolved oxygen from the water flowing over the gill filaments). Dissolved carbon dioxide is what marine plants use for photosynthesis.
The amount of dissolved gases varies according to the types of life forms in the water. Most living species need oxygen to keep their cells alive (both plants and animals) and are constantly using it up. Replenishment of dissolved oxygen comes from the photosynthetic activity of plants (during daylight hours only) and from surface diffusion (to a lesser extent). If there are a large number of plants in a marine water mass then the oxygen levels can be quite high during the day. If there are few plants but a large number of animals in a marine water mass then the oxygen levels can be quite low. Oxygen is measured in parts per million (also called ppm) and levels can range from zero to over 20 ppm in temperate waters. It only reaches 20 when there are a lot of plants in the water, it is very sunny with lots of nutrients, and the wind is whipping up the surface into a froth. In any water mass there is a maximum amount of dissolved gas that can be found (after which the gas no longer dissolves but bubbles to the surface). This maximum amount increases with a decrease in temperature (thus cold water masses can hold more dissolved gases … but they can also have none if it has been used up). So, just because a water mass is cold it does not mean it has a lot of dissolved gases. This concept is a little tricky but just remember that the amount of dissolved gases in seawater depends more on the types of life forms (plants and animals) that are present and their relative proportions.
pH
pH is a measure of the acidity or alkalinity of a substance and is one of the stable measurements in seawater. Ocean water has an excellent buffering system with the interaction of carbon dioxide and water so that it is generally always at a pH of 7.5 to 8.5. Neutral water is a pH of 7 while acidic substances are less than 7 (down to 1, which is highly acidic) and alkaline substances are more than 7 (up to 14, which is highly alkaline). Anything either highly acid or alkaline would kill marine life but the oceans are very stable with regard to pH. If seawater was out of normal range (7.5-8.5) then something would be horribly wrong.
Ref http://www.marinebio.net/marinescience/02ocean/swcomposition.htm
ACIDITY REDUCED ALKALINITY:
“Today, the surface waters of the oceans have already acidified by an average of 0.1 pH units from pre-industrial levels, and we are seeing signs of its impact even in the deep oceans”, said co-author Dr. Eva Calvo, from the Marine Science Institute of CSIC in Barcelona, Spain.
Ref http://wattsupwiththat.com/2010/03/30/ocean-acidification-the-evil-twin-of-global-warming/
ARC Centre of Excellence for Coral Reef Studies Ref http://www.coralcoe.org.au/news_stories/acidocean.html
INCREASED CO2 BENEFITS:
A recent paper in Geology (Ries et al., 2009) found an unexpected relationship between CO2 and marine calcifers. 18 benthic species were selected to represent a wide variety of taxa: “crustacea, cnidaria, echinoidea, rhodophyta, chlorophyta, gastropoda, bivalvia, annelida.” They were tested under four CO2/Ωaragonite scenarios:
409 ppm (Modern day)
606 ppm (2x Pre-industrial)
903 ppm (3x Pre-industrial)
2856 ppm (10x Pre-industrial)
7/18 were not adversely affected by 10x pre-industrial CO2: Calcification rates relative to modern levels were higher or flat at 2856 ppm for blue crab, shrimp, lobster, limpet, purple urchin, coralline red algae, and blue mussel.
6/18 were not adversely affected by 3x pre-industrial CO2: Calcification rates relative to modern levels were higher or flat at 903 ppm for halimeda, temperate coral, pencil urchin, conch, bay scallop and whelk.
3/18 were not adversely affected by 2x pre-industrial CO2: Calcification rates relative to modern levels were higher or flat at 903 ppm for hard clam, serpulid worm and periwinkle.
2/18 had very slight declines in calcification at 2x pre-industrial: Oyster and soft clam.
The effects on calcification rates for all 18 species were either negligible or positive up to 606 ppm CO2. Corals, in particular seemed to like more CO2 in their diets…
Ref http://wattsupwiththat.com/2011/01/10/ocean-acidification-chicken-of-the-sea-little-strikes-again/
OPINION requiring CONFIRMATION:
It is clear that human-induced changes in atmospheric CO2 concentrations are fundamentally altering ocean chemistry from the shallowest waters to the darkest depths of the deep sea. The chemistry of the oceans is approaching conditions not seen in many millions of years, and the rate at which this is occurring is unprecedented (Caldeira & Wickett 2003).
Caldeira and Wickett (2003, p. 365) state “Unabated CO2 emissions over the coming
centuries may produce changes in ocean pH that are greater than any experienced
in the past 300 million years, with the possible exception of those resulting from rare, catastrophic events in Earth history” (Caldeira and Rampino 1993; Beerling and Berner
2002).
Ref http://www.gg.mq.edu.au/rep/websites/docs/paper.pdf

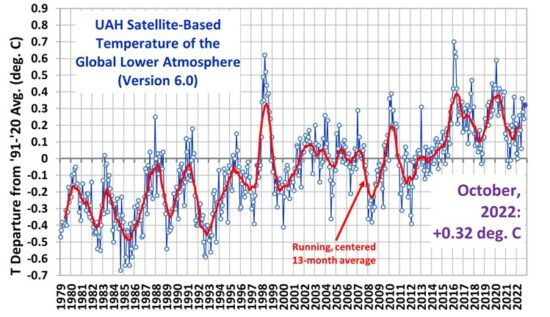




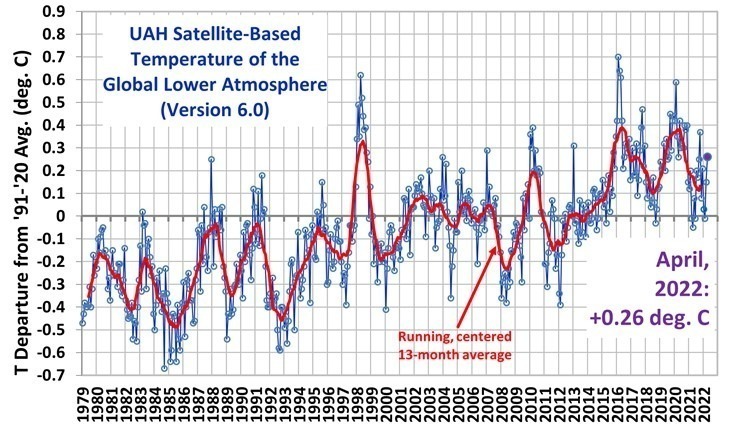


A very interesting blog. Well researched and laid down.
I consume very little shellfish, hardly any. Not for some years now. Still the state of our oceans is of concern for me. The mercury and PCB pollution. Of Salmon and Tuna is another concern, also. As people just chuck garbage into the oceans, as if it were a giant garbage dump. All those boats, just dump their waste over the side. … etc. Killing sharks, for their fins, then throwing the bodies back. Little wonder the balance of ocean life is changing? Over fishing often by drag nets causing major disruption to ocean floor. Rays and other bottom feeders. As we keep hearing about the benefit of consuming fish, for omega 3, etc. One of the best sources for humans, is hemp. http://www.pkdiet.com/pages/herbs/food/hempseed.htm Pointed out in one of your recent blogs.
The city of Victoria in B.C. just dumps it’s waste offshore. You should see the fields of bull kelp, it produces. The proposal to build a water treatment plant, is a hot button issue there. Yet it is a vey wealthy city. Maybe somewhat appropriate? The relationshop between s**t and c**p?
Most people seem to go through life blissfully unaware? They do not care as long as it does not directly affect them. They fail to see their quality of life is slowly dwindling. Just suck it up, excrete it out and carry on.